Since the start-of the Covid-19 pandemic, there has been an enormous surge in demand for Covid-19 testing, which has taken us back to an interview with Michela Savoldi-Boles, Chief Technology Officer of Bioside Diagnostics, earlier this year, who talked about the challenges of rapid scale-up during an extremely challenging time in the Lombardy region of Italy, and on Covid-19 testing accelerated with Andrew+.. read on..

Q: Please could you tell us a little bit about Bioside Diagnostics and your role there
A: Bioside is a young, Italian Company, operating in molecular biology and microbiology. We have our HQ in Lombardy in the North of Italy. We are located in a biotechnology centre, and have benefitted greatly from collaborations and networking due to the number of research centres, start-ups and other institutes, especially during the early days of our company. I am Chief Technology Officer of Bioside and I am a biologist, responsible for the development and manufacturing of the Bioside kits. Our Company produced and developed molecular biology kits and microbiology kits especially in the environmental and food fields, and this year we also started to produce kits for the clinical and diagnostic areas. We have been on the market for 5 years and during this time had a lot of collaboration with major players such as Thermo Fisher Scientific.. actually we produce kits for Thermo and we have other distributors on the market because we are now approaching the global market for the first time.
We have been performing clinical tests in our laboratory since March 2020. We are also producing the test kits but in order to give support to our region especially, because Lombardy was the first region to be affected in Italy, our region needs to have more laboratories in order to be able to do the number of tests required, so we also perform the required analytical work. It is a temporary situation in order to give our support to the state.
Q: I understand that Bioside is developing a diagnostic test for COVID-19. Please could you tell us a little bit about the test and how it works.
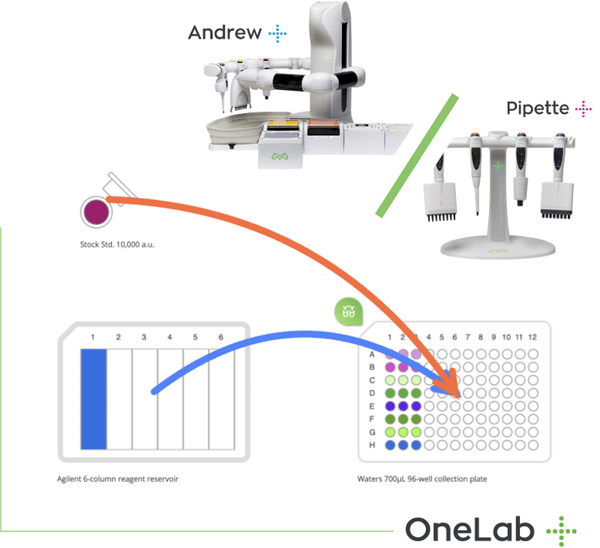
A: We produced kits in molecular biology with a proprietary technology , ensuring that they are stable at room temperature and are ready to be used by the customer. The technology is based upon Real Time PCR and the production technology is proprietary and patented by Bioside. The innovation for our customers is that for them it is very easy to use because all the reagents are pre-dosed and dispensed direct into the plastics so the customer needs only to put the RNA or DNA into our test tubes and then perform the analysis, so it is very simple to use. The reagents are stored at room temperature for both storage and for transport. We also have a technology for processing both the RNA and the DNA so when COVID-19 occurred we had the perfect combination of technologies in order to develop a kit for SARS COVID-19. It was very difficult to develop these kits in February because there was has been limited information about the virus and the situation very different from our standard practice. A further innovation is the fact that our kits also detect the presence of different mutations of SAR COVID-19, in order to give the best technical specification of the product for our customers.
Q: What are the key challenges in developing the test and how has working in lockdown, in Italy, affected that?
A: It was very difficult as during the lockdown, we needed to be open so that we could continue to operate as well as to produce kits. We provide diagnostics tests for application in both hygiene assessment and the clinic. During lockdown, the test results were urgently required but in order to respect social distancing we needed to deliver faster results with less staff. We needed to operate from 7am to 7pm every day of the week. We had problems with some of our suppliers as some were COVID contaminated, and we needed to carefully check materials and cells received in order to verify that the virus was not present. This was especially challenging as the situation changed each week. For example, regulations kept changing, and when some of our staff fell ill, we needed to replace them at short notice.
Q: How do you see our automation (Andrew+) supporting your development work?
A: It was very important because during the rapidly evolving situation we needed to upgrade our production capacity from 100 tests to 1000 tests per day so we needed to automate some of our processes, especially those involving liquid handling. Achieving this whilst needing to reduce staff was very difficult.
We have had experience with other automation platforms and found that, in some cases, the automation is not flexible. We chose Andrew Alliance as the Andrew+ system was more flexible than others and we saw it better fitted our needs, especially given the circumstances where we couldn’t predict what might happen tomorrow. Its highly flexible workspace enabled us to quickly adapt our workflow. Covid-19 testing accelerated with Andrew+. This is why we chose Andrew+.

Andrew+ liquid handling robots being used under laminar flow hoods.
The set-up of the robot was very rapid and very simple.. we have experience of other instruments that need days to do the installation and a lot of technical support.. instead with Andrew+, the instrument was ‘ready to go’ and just needed 2 hours of remote support in order to complete installation and to have it up and fully working..
..the ability of the Andrew personnel during the installation was very punctual and rapid because they are available by phone, by internet, by email.. we felt very supported by Andrew Alliance during the installation..
Q: What aspects of both Andrew+ and the OneLab software do you see most beneficial.. and why?
A: The flexibility during the program and execution and running of protocol and also the flexibility in the use of different Dominos as well as the number of Dominos.. in order to change the application of Andrew+, you just need to swap Dominos. Covid-19 testing accelerated with Andrew+.
Our test is now on the market (since May).. it requires the correct certification ‘marks’ in order to permit its use in hospitals. We are working on gaining European authorisation for use in an emergency.. now we are distributing the tests to hospitals as well as for laboratories involved in environmental and clinical testing.
Learn more at www.andrewalliance.com