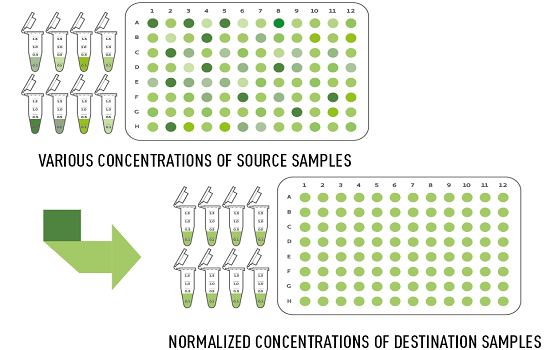
Concentration normalization
For the tedious and error-prone concentration normalization task, the user-friendly software Andrew Lab and the compact Andrew robot provide time and cost saving solutions. Hands-on time is reduced by 16 times while liquid transfer reproducibility is improved by 1.5 times with respect to an experienced human operator.
Concentration quantification and normalization are essential at the start of many genetic, genomic, and proteomic applications, such as NGS library preparation, qPCR, q-RT-PCR, QC of protein and antibody production, genotyping, sequencing, clinical diagnostics, forensics, etc. Manual pipetting during normalization requires very tiresome changing of pipet volumes for both sample and diluent, especially i
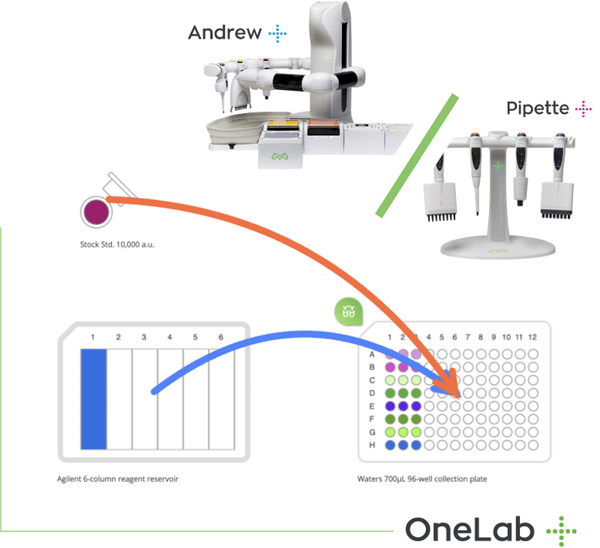
Figure 1: Simplified normalization procedure by Andrew Lab and the robot Andrewn medium-throughput applications using microplates. Mistakes easily occur during the calculation of the required transferred volumes and by dispensing the wrong liquid amount into the wrong destination. Furthermore, performance variations within and between human operators are unavoidable and create additional non-biological errors in downstream steps, requiring costly rework of the entire process.
All of these mistakes and pain can be completely avoided by using the Normalization wizard in the software Andrew Lab together with the robot Andrew (Figure 1). All that is needed is the user’s spreadsheet containing the sample location with the original concentrations and volumes – either in multi-well plates or set of tubes. A few simple clicks in Andrew Lab will generate the complete pipetting protocol, automatically calculating the buffer and sample volumes needed to achieve an equal concentration in all sample. After providing your samples and buffer on the working deck of Andrew, a simple “Start” button will initialize the robot to execute the normalization protocol, without any mistakes, from any source to any destination of common labware such as microtubes, multi-well plates, tubes or vials. The resulting normalized samples are then ready to use for immediate downstream applications, with absolute certainty on their correct concentration. All individual pipetting steps and intermediate liquid concentrations can be easily traced at any step, or exported for documentation conforming to the Good Laboratory Practice guidelines.
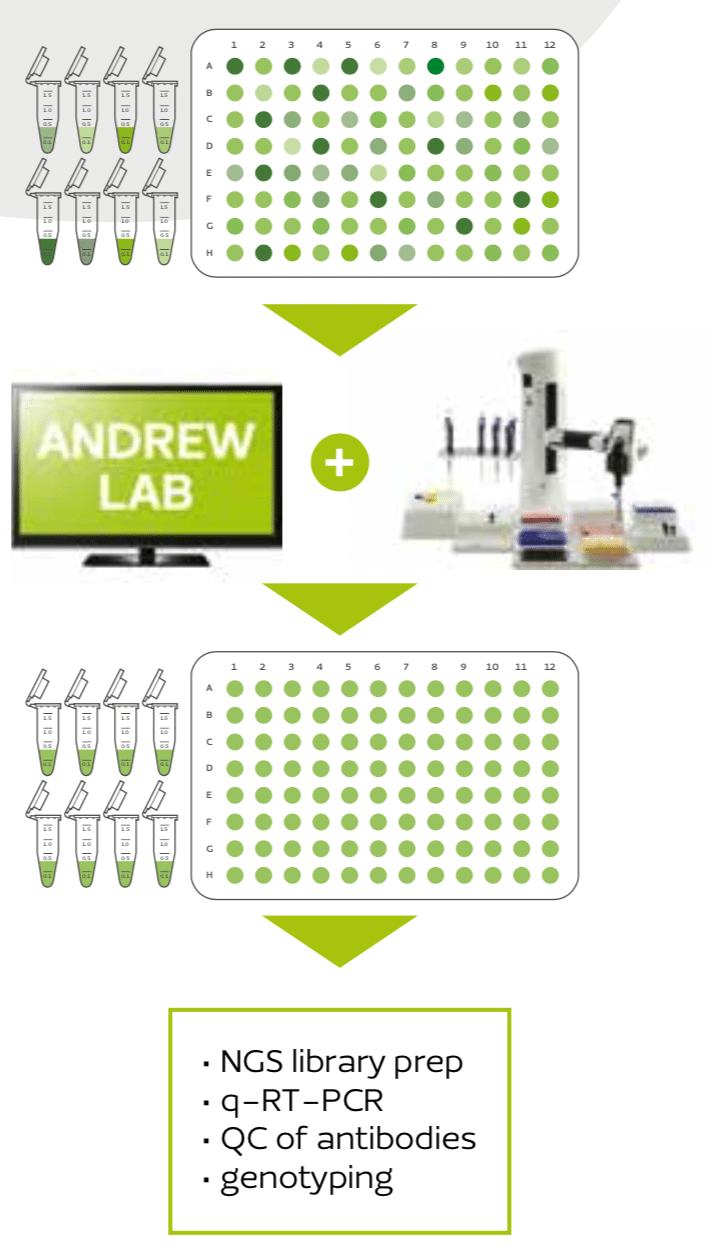 Figure 1: Simplified normalization procedure by Andrew Lab and the robot Andrew
Figure 1: Simplified normalization procedure by Andrew Lab and the robot Andrew
Andrew decreases hands-on time by 16 times and improves reproducibility by 1.5 times with respect to an experienced human operator
The normalization performance of five Andrew robots (four 1000G and one 1000R model) with three different Gilson pipette sets on different days were evaluated by the photometric assessment method with the Ponceau S dye. Each Andrew was programmed to (a) first, generate 96 dye solutions at 96 different concentrations (18.5 – 95 mg/L) by mixing distilled water with a dye stock solution (100 mg/L) in a 96-well plate to achieve 250 μL, and (b) normalize these 96 dye concentrations into ano-ther 96-well plate to the same concentration of 18 mg/L for a volume of
150 μL. The volume range transferred by Andrew was 12.5 – 237.5 μL, using the four pipettes P20, P100, P200, and P1000 and Gilson’s Diamond tips.
One Andrew 1000G was tested for the reproducibility of creating 96 different concentrations by 4 replicated plates. The CV values computed for each of these 96 concentrations from these four plates show the majority of the samples being uniform within 1%, indicating excellent p petting reproducibility (Figure 2)
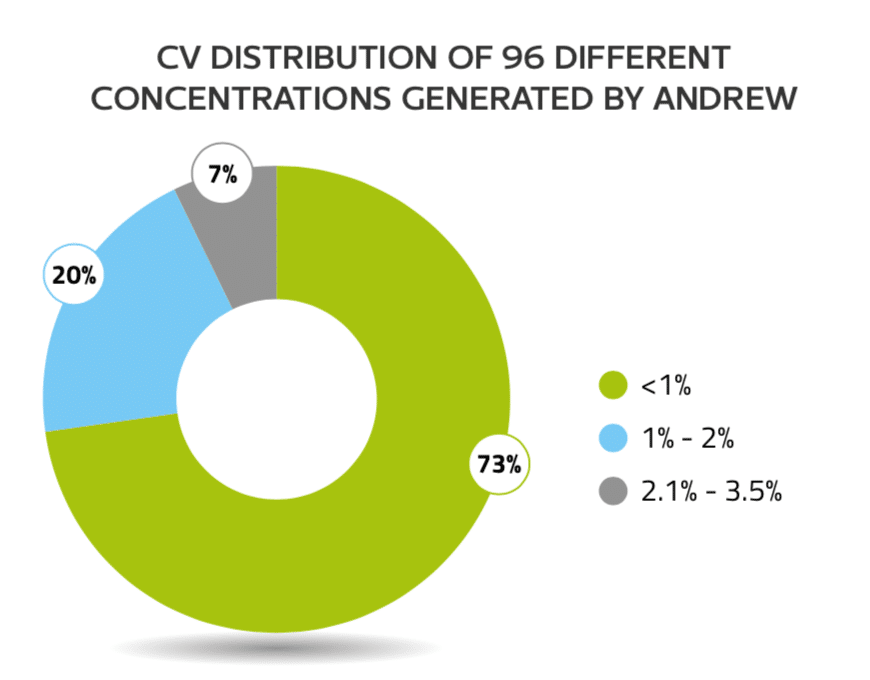
Figure 2: Andrew reproducibly created 96 different concentrations
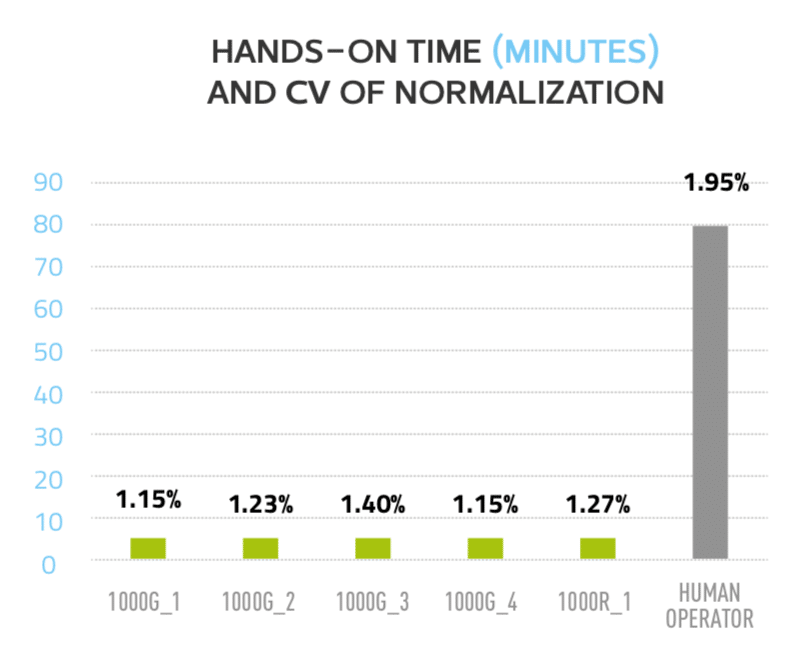
Figure 3: Andrew required significantly less hands-on time while improving reproducibility of normalization
Five different units were then tested for the reproducibility of normalizing 96 different concentrations in a source plate into another destination plate at one final concentration. For each Andrew, the CV of these 96 normalized concentrations was computed. Automatic pipetting by Andrew was compared with the data generated by a human operator with 18 years of manual pipetting experience repeating the same normalization procedure, and using the same pipette set. All five Andrews performed with better and uniform CVs (1.15% – 1.4%) significantly improving the results from a human operator (1.95%) (Figure 3).
The user’s total hands-on time for the entire procedure using Andrew Lab and the robot Andrew was 5 minutes, including 2.5 minutes of designing the protocols and 2.5 minutes of arranging the samples and buffer onto Andrew’s bench space. On the other hand, the manual normalization procedure took ~ 1h20m of hands-on time for the human operator. Although each Andrew completed the normalization protocol in ~1h30m, the hands-off time for the user was improved by 16 times since Andrew works fully unattended (Figure 3).
Simplified designing of normalization protocols in Andrew Lab
Starting with the spreadsheet of the sample concentrations sorted from highest to lowest or vice versa, users can just copy and paste this sample list into Andrew Lab. This sorting will expedite the execution of Andrew as less time is required for changing volumes and pipettes. Sample volumes and positions in the source consumable are also required in this list. Within 2 min, any normalization protocol will be generated by the Normalization wizard of Andrew Lab in 6 simple steps executed in a few clicks by the user.
Step 1: Copy and paste the source names, their locations, and the initial concentrations and volumes.
Step 2: Indicate the consumables containing the samples to inform the wizard of the input format (Figure 4).
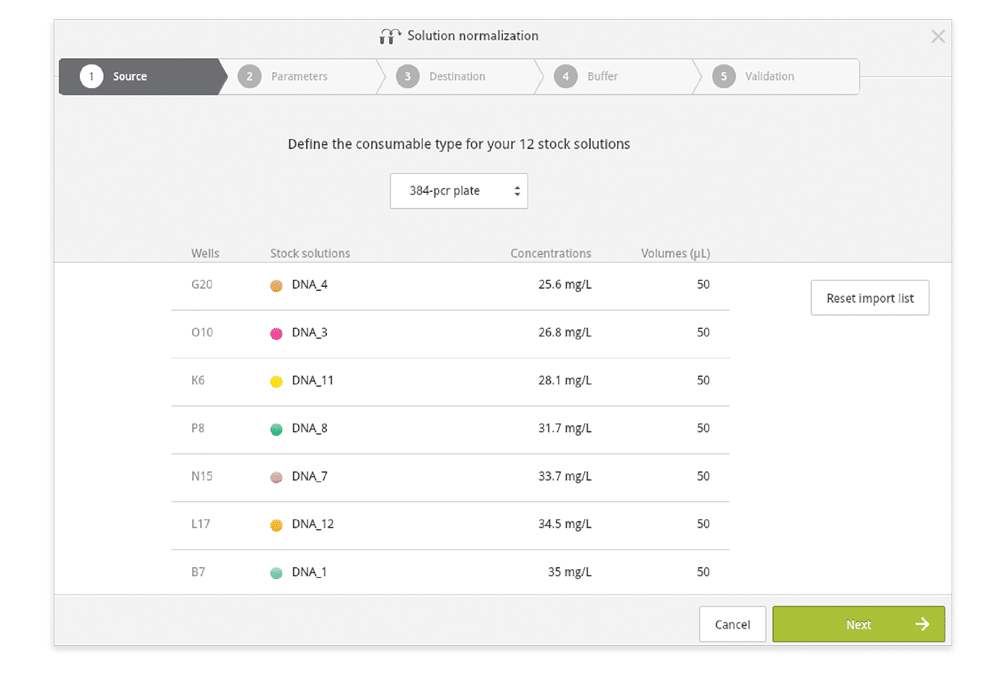
Figure 4
Step 3: Select the normalization method as either: (A) at a fixed volume in different destinations (Figure 5A), or (B) at variable volumes in the same source (Figure 5B). For both cases, indicate the desired normalized concentration. Please remark that the wizard will know the practical limits that are physically possible depending on the source and destination capacity.
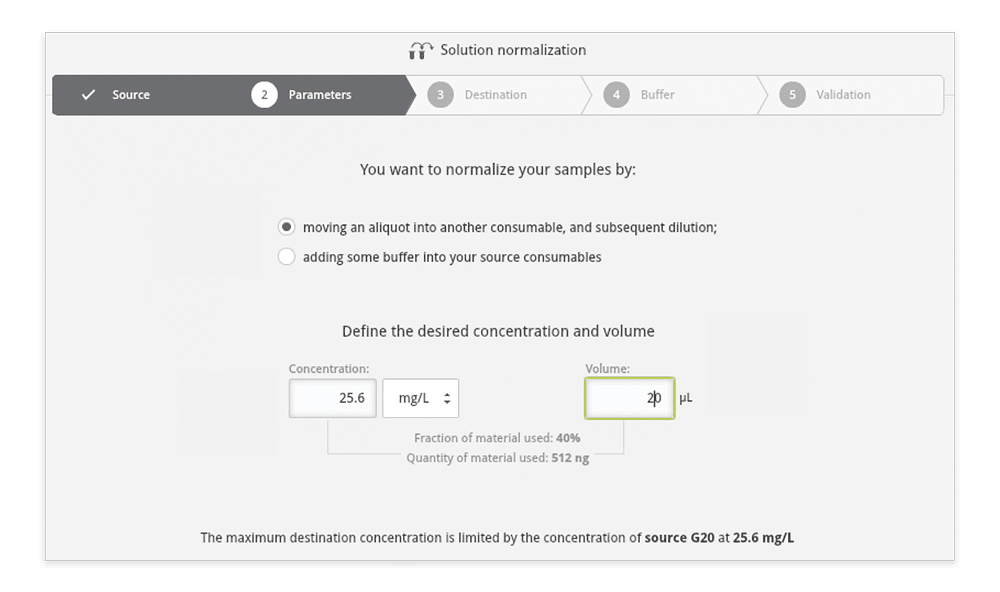
Figure 5A

Figure 5B
Step 4: If method 3(A) is selected, choose the destination consumable type and positions (Figure 6); if method 3(B) is selected, the wizard skips to Step 4 because the destination consumable is the same as the source consumable.
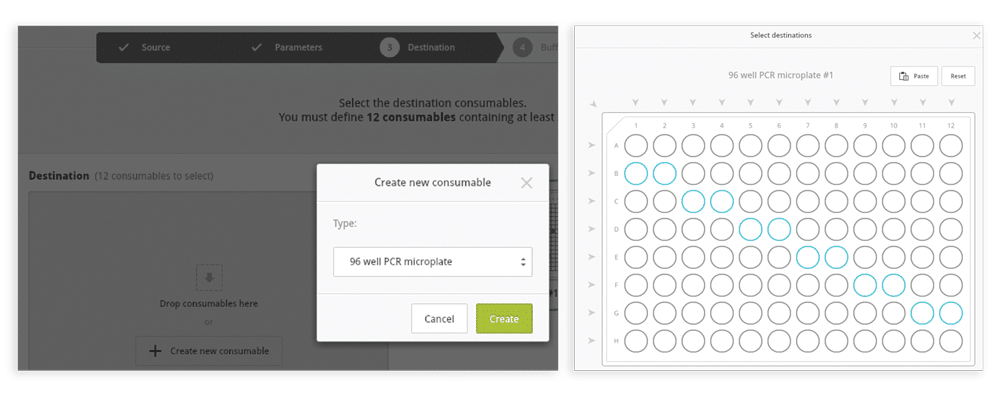
Figure 6
Step 5: Indicate the Buffer for diluting the samples (Figure 7).
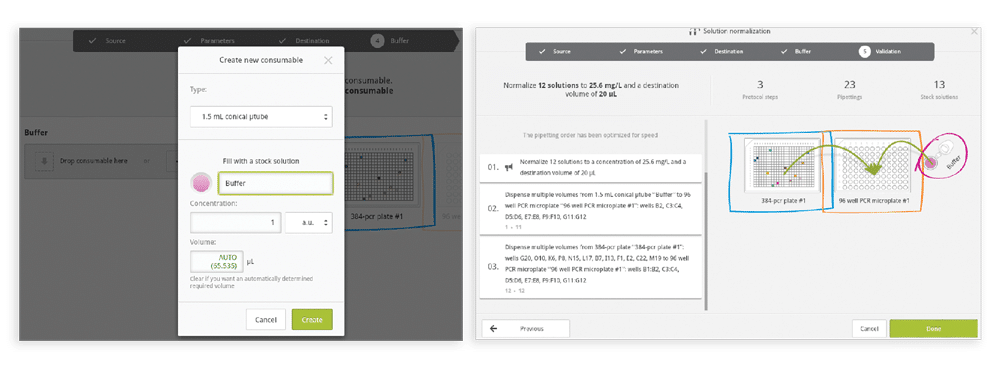
Figure 7 & 8
Step 6: Validate and accept the newly created normalization pro- tocol (Figure 8). Once the normalization protocol is generated, the pipetting steps and transferred volumes of the buffer and samples calculated by Andrew Lab can then be verified as standard pipetting actions within Andrew Lab. For example, you can: • Verify protocol steps (Figure 9)
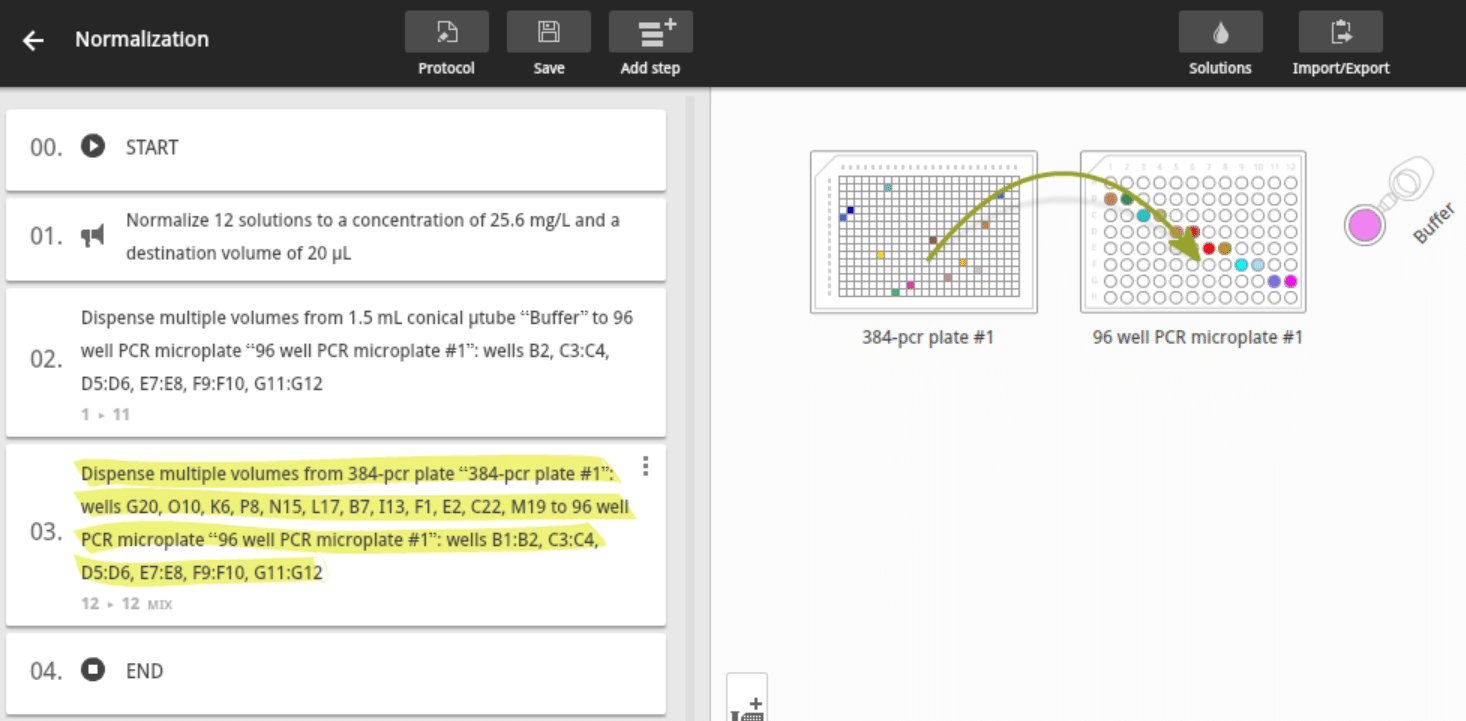
Figure 9
- Verify the transferred sample volumes (Figure 10):
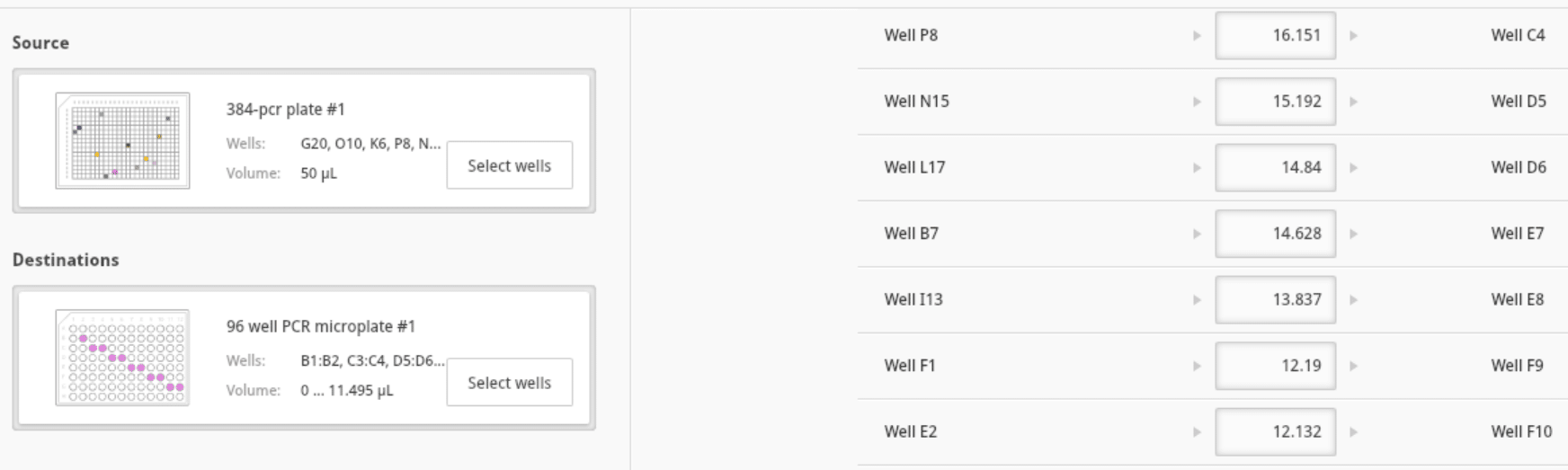
Figure 10
Users can also additionally modify other protocol parameters such as tip choice, tip positions, and mixing at the source and destinations. For more details on how to use the Normalization wizard of Andrew Lab, users are referred to the Andrew lab manual.
Summary
As a tireless companion and assistant to technicians and scientists, the pipetting robot Andrew will transform the tedious procedure of concentration normalization into a painless process for users, providing many benefits:
• Accurate and reproducible pipetting in complex patterns with systematic and random errors below that of human operators, thus improving quality of work flows
• Automatic pipetting significantly decreasing risks of repetitive strain injury of human workers
• Automatic calculation of the complex list of dilution volumes directly in Andrew Lab and delivery of a ready-to-execute normalization protocol, without possibility of protocol design errors.
• Wide compatibility with diverse lab consumables in large volume range of 0.2 μl – 10 mL, using user existing samples and providing ready-to-go formats for immediate downstream applications.
• Decreasing hands-on time of users by 16 times for each normalization process, thus enabling multi-tasking and enhancing lab productivity.
With only five minutes of total hands-on time to design the most complex normalization protocols, users can literally walk away for other value-added activities, assured that all their samples are normalized accurately and reproducibly with performance superior to that of the most expert human operators.